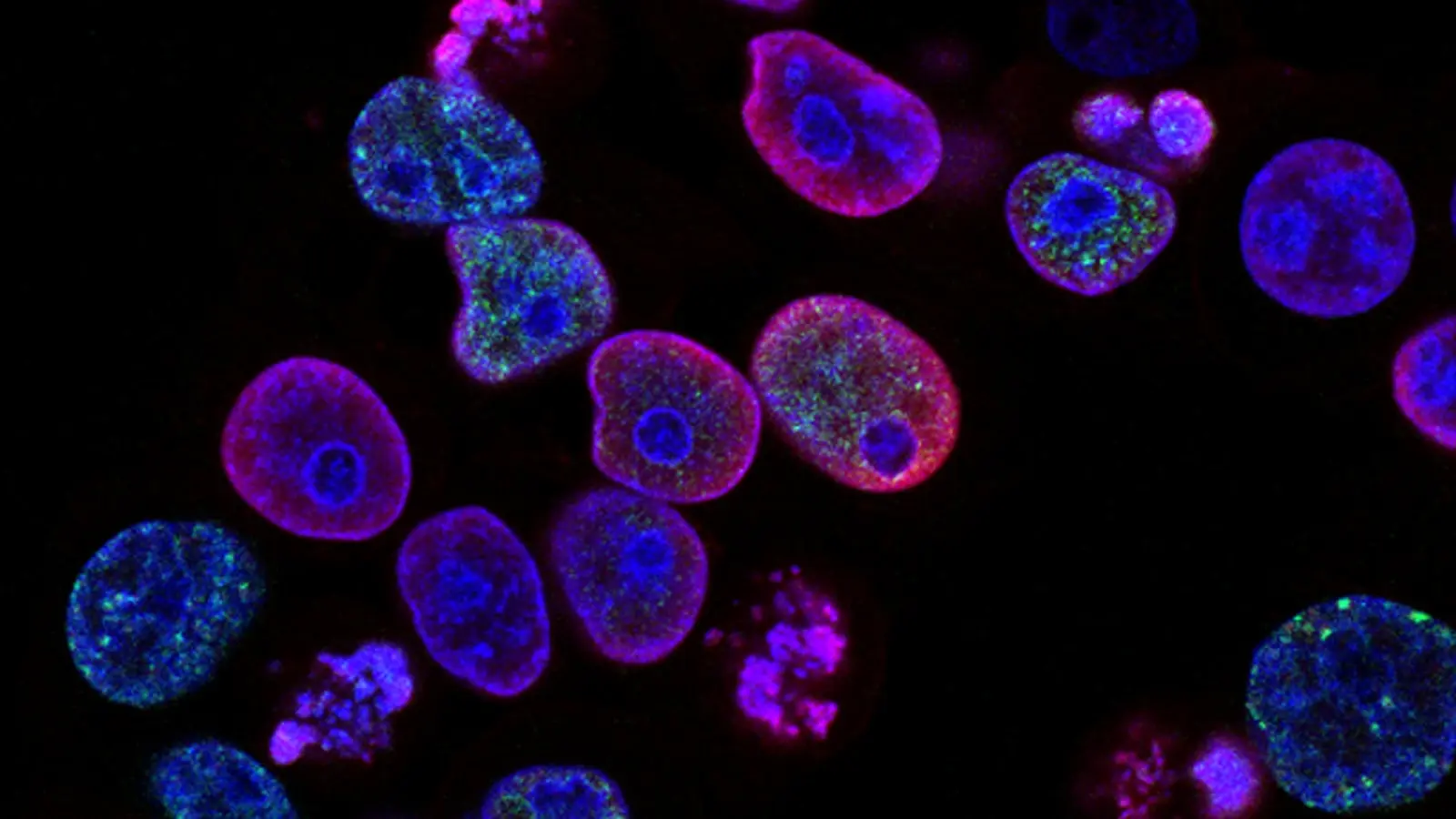
Researchers have located a single point of failure in the malaria parasite’s life cycle, a protein that acts as a master switch for its replication and transmission. The discovery of this molecular linchpin, named Aurora-related kinase 1 (ARK1), provides a precise target for a new generation of antimalarial drugs, a critical development as existing treatments falter against evolved resistance. When scientists genetically disabled ARK1, the parasite, Plasmodium falciparum, was rendered inert, unable to complete its development in either human or mosquito hosts. The transmission cycle was broken.
This is not a minor disruption. It is a complete halt. The research offers a promising roadmap in a fight where the biological landscape is constantly shifting underfoot. For decades, medicine has been chasing the parasite as it evolves resistance. Identifying a fundamental, non-negotiable component of its survival mechanism allows for a more proactive strategy. The pressure to find such a target is immense, with hundreds of thousands of lives, mostly children in sub-Saharan Africa, lost to the disease each year. The relentless spread of artemisinin resistance across the continent has turned the search for novel drug targets from an academic exercise into an emergency.
The Molecular Kill Switch
The parasite’s machinery grinds to a halt without ARK1. As a kinase, its job is to chemically tag other proteins, signaling them into action. Specifically, ARK1 orchestrates the formation of the mitotic spindle, the delicate scaffolding that pulls duplicated chromosomes apart during cell division. Without this scaffold, the parasite cannot multiply. It is a fundamental process. In labs, researchers watched as parasites with deactivated ARK1 failed to divide, their growth arrested inside the host’s red blood cells. They became biological dead ends.
The effect was comprehensive. The parasite not only failed to replicate in the human bloodstream but was also unable to complete the sexual development stage required for transmission back to a mosquito. This double failure makes it an exceptionally powerful target, breaking the cycle of infection at two distinct points. A parasite that cannot spread is a parasite that can be eradicated. “The name Aurora refers to the Roman goddess of dawn,” noted Dr. Ryuji Yanase of the University of Nottingham, whose team led the research. “We believe this protein truly heralds a new beginning in our understanding of malaria cell biology.”
A Target Without Collateral Damage
The strategic value of ARK1 is amplified by its absence in humans. The protein has no direct human equivalent, or ortholog, making it an ideal candidate for therapeutic intervention. Drugs designed to inhibit ARK1 could, in principle, attack the parasite with surgical precision, leaving human cells unharmed. This avoids the collateral damage that complicates many drug therapies and reduces the likelihood of severe side effects. (The gap between identifying a target and delivering a viable drug, of course, remains vast).
The challenge now shifts from biology to chemistry: designing a small molecule compound that can penetrate a human red blood cell, find the parasite hiding within, and selectively block the ARK1 protein. It is a lock-and-key problem on a nanoscale, played out in the complex environment of the human body. Finding a compound that is stable, effective, and safe is the next great hurdle. The discovery provides the coordinates; the pharmaceutical industry must now build the missile.
A Multi-Front War
The discovery of ARK1’s vulnerability does not exist in a vacuum. It is one advance in a rapidly accelerating, global campaign against malaria. This is a biological arms race fought in laboratories, clinics, and with AI data models. In parallel, Brazilian researchers are testing a synthetic compound with the potential to treat all three stages of the malaria life cycle simultaneously—a brute-force approach compared to the ARK1 precision strike. Elsewhere, AI-powered mapping tools have charted over 20,000 protein interactions within the parasite, creating a vast database of potential new weaknesses. The sheer volume of data is changing how these vulnerabilities are found.
And on the front lines, a new drug called GanLum, tested across 12 African nations, has demonstrated over 97% effectiveness against artemisinin-resistant strains. For every 100 patients treated, 97 are cleared of the parasite. These efforts represent the current war. The ARK1 discovery opens a blueprint for the next one. It signals a shift from adapting old weapons to engineering new ones based on an intimate knowledge of the enemy’s biology.
The identification of ARK1 provides more than just another potential drug target; it offers a high-resolution map of the parasite’s core machinery. By targeting a fundamental, non-redundant component of its survival, science is shifting from chasing the parasite to cornering it. The dawn protein is not a cure. It is a critical vulnerability. And it is now exposed.